Clinical Research Explained
Research and clinical trials are an everyday part of the work done in the NHS.
Research doesn’t always involve staying or visiting hospital or a GP practice - important research can be done without leaving home, or can be supported by giving your permission for spare blood from routine tests to be used for research.
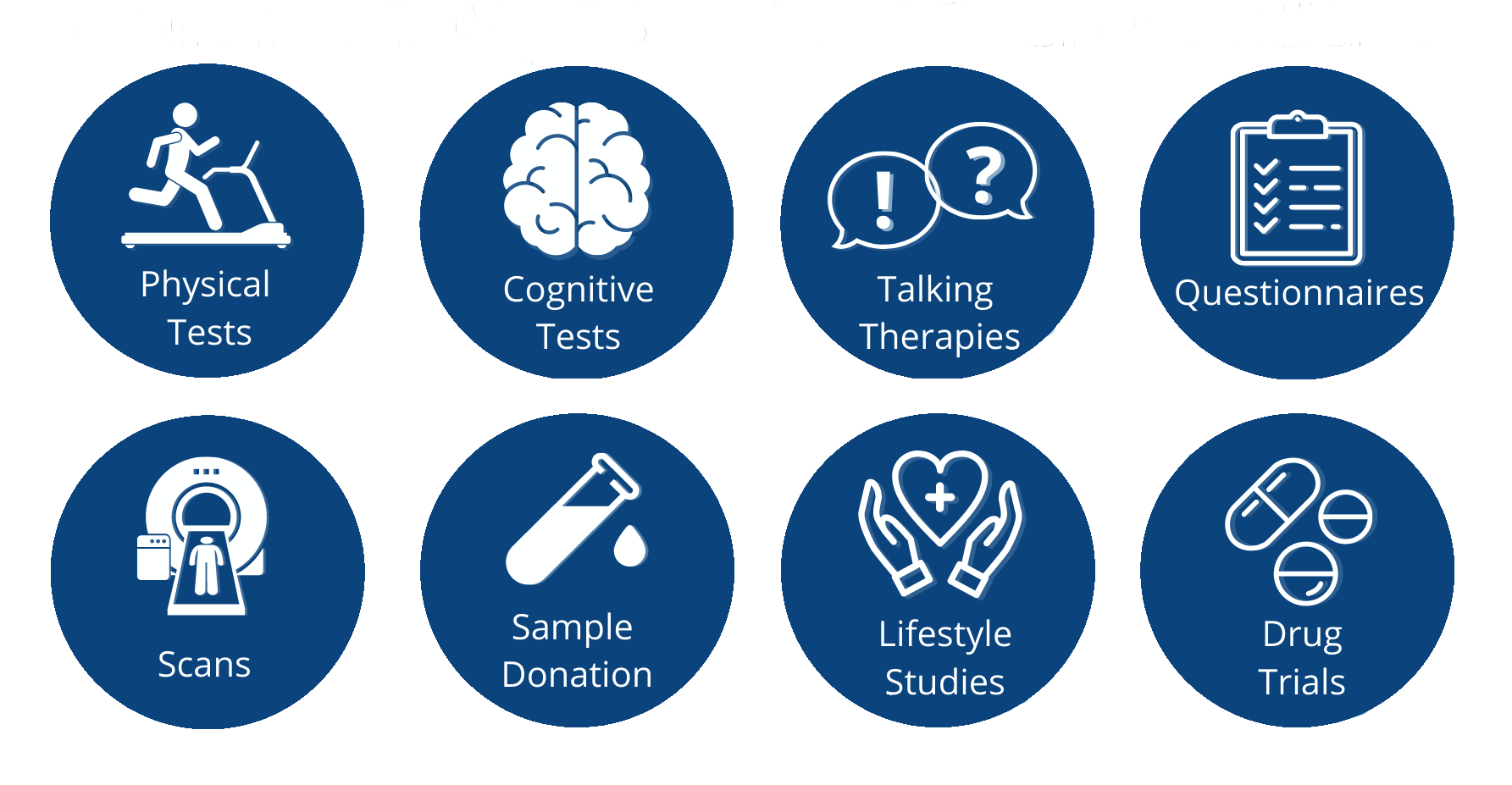
Common types of research include:
Before treatments can be tested in humans, they will have already undergone extensive investigation. The researchers conducting the trial must draw up a plan or protocol which will include information such as:
- who and how many will take part in the trial
- what question does the trial aim to answer
- what treatments will be compared
- how the results will be collected
The protocol then undergoes an independent scientific review and must also be approved by an ethics committee before the trial can go ahead. These checks are to ensure that the trial design is accurate and that the research will respect the rights, dignity, safety and wellbeing of the participants.
Involvement of patients, carers and the public are vital at each stage with opportunities to:

